The efficacy of LOLA has been investigated in various clinical studies. In the studies described here the use of LOLA (infusion, oral administration or a combination of the two) was documented in patients with mild to severe liver insufficiency. Cirrhosis was the most often underlying cause of hepatic dysfunction, although patients with other liver diseases were also treated. Studies varied from a few days to several years in duration.
Measurement of ammonia concentration has been included in clinical trials as a key feature on the mechanism of action. A close relationship has been demonstrated between changes in the ammonia concentrations on treatment with LOLA and the clinical therapeutic efficacy.
Effects of LOLA on ammonia concentration1
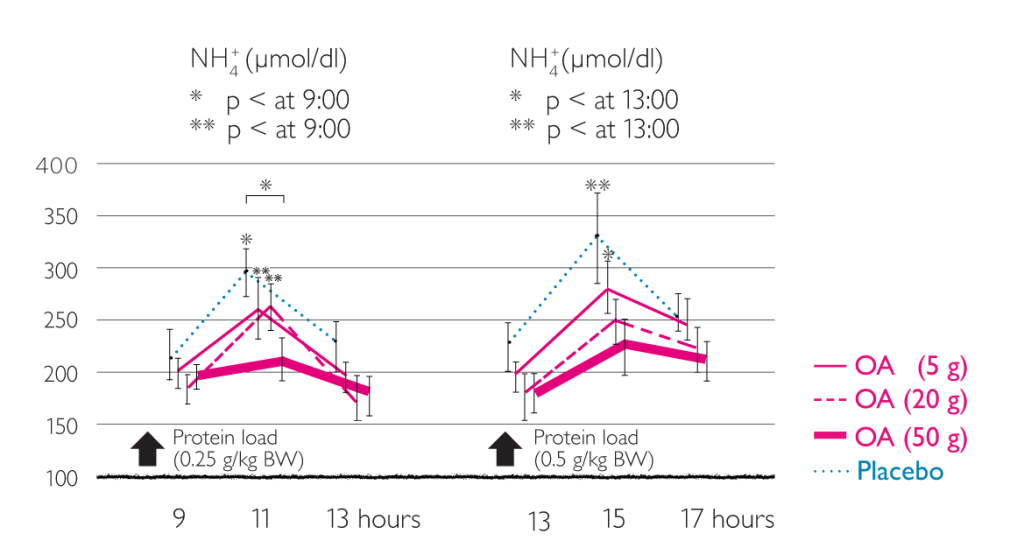
Lowering of ammonia by intravenous LOLA1
Effect of LOLA on protein synthesis in muscle2
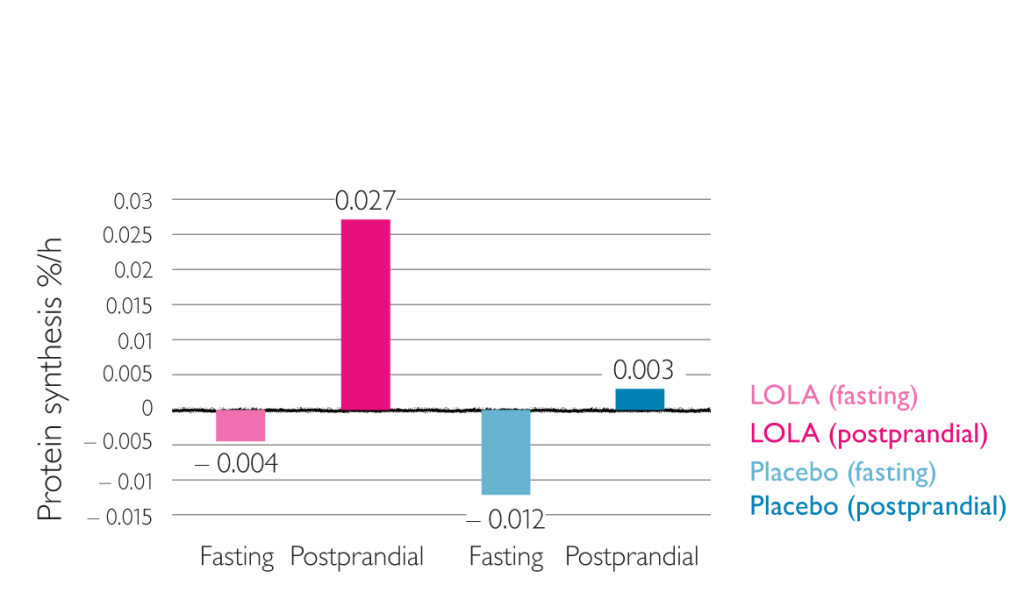
Results of this study show inhibition of catabolic muscle metabolism and stimulation of protein synthesis in cirrhosis patients by LOLA2
Effect of LOLA on brain amino acids3
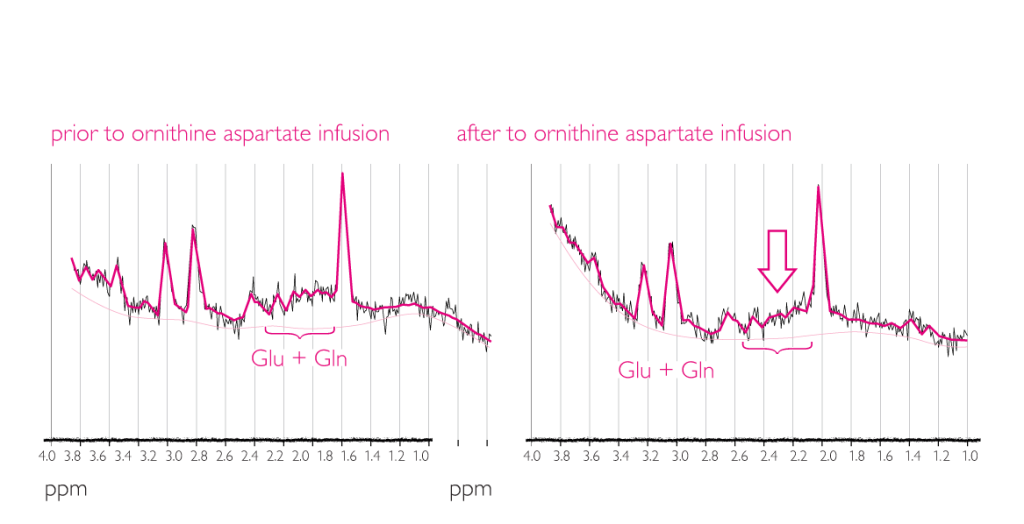
This study demonstrated a decrease in glutamine and glutamate concentration with LOLA3
Efficacy of Hepa-Merz® infusion
Therapeutic efficacy of L-ornithine-L-aspartate infusions in patients with cirrhosis and hepatic encephalopathy: results of a placebo-controlled, double-blind study. Kircheis G et al. (1997).4
This was a placebo-controlled, double-blind trial on 126 patients with cirrhosis of the liver and HE grade 0 to II. All patients had to have demonstrable hyperammonemia (venous ammonia concentration >50 µmol).
Treatment with LOLA was associated with significant improvements including:
- Lowering of ammonia concentrations under fasting conditions
- Decreasing postprandial ammonia concentrations
- Highest improvement in patients with HE grade ii, but also with sub-acute HE and grade I
- Improvements in PSE index
Significant improvements in patients with cirrhosis, HE and hyperammonemia treated with LOLA infusion4
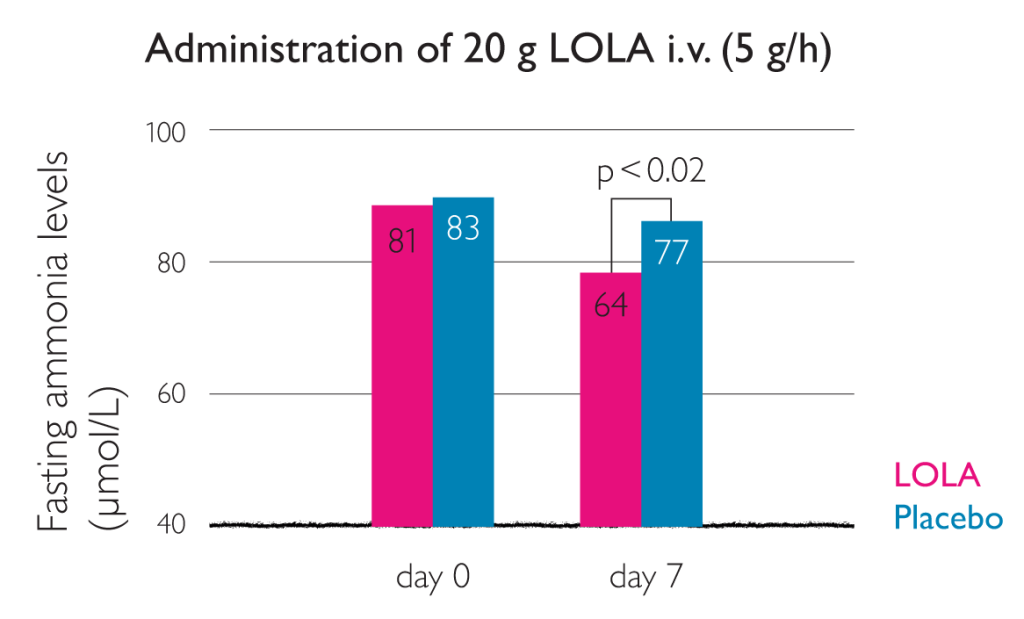
This graph shows that LOLA administration lowers ammonia levels under fasting conditions4
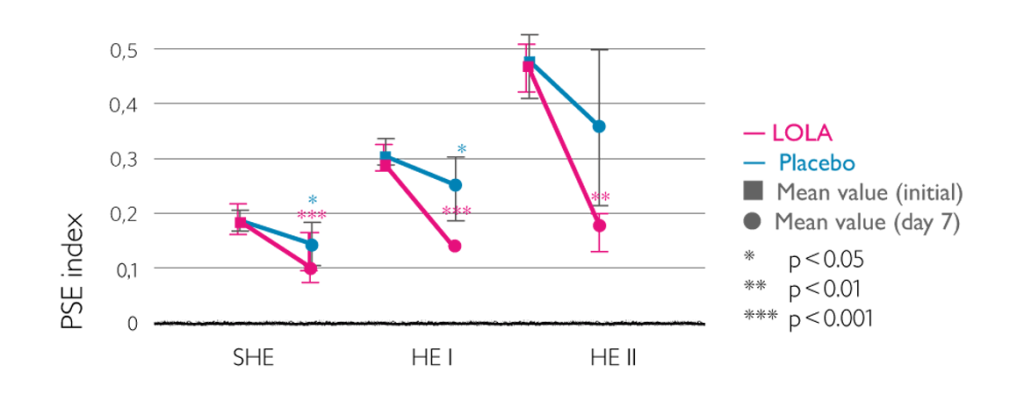
Improvement in PSE index as result of intravenous LOLA4
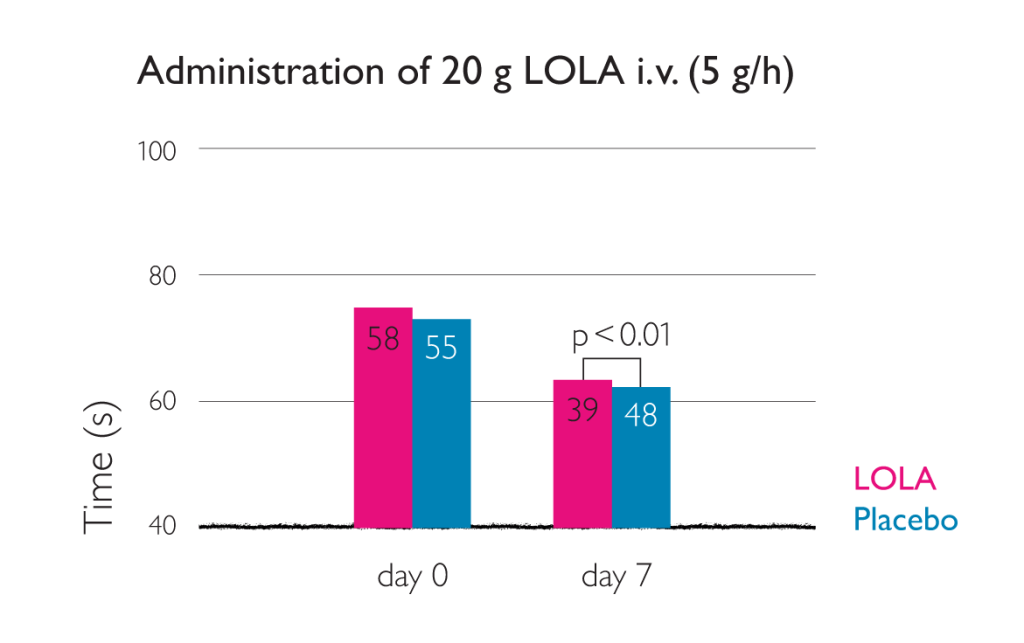
Time taken for NCT-A test reduced by intravenous LOLA4
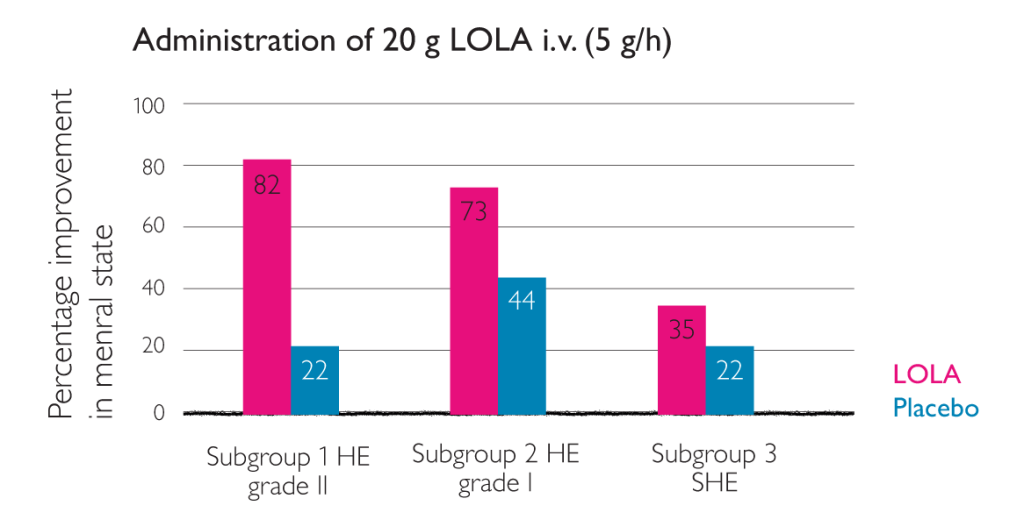
Improvement in HE as a result of intravenous LOLA4
Therapeutic effect of LOLA on liver cirrhosis complicated by hepatic encephalopathy. Chen MF et al. (2005)5
Eighty-five patient with liver cirrhosis complicated by hepatic encephalopathy were divided into therapy group (n=45) and control group (n=40). Patients in the control group were treated with routine comprehensive therapy, and those in the therapy group received additional intravenous administration with 40 ml LOLA in 250 ml 10% glucose and saline (once daily, 7 days for a treatment course).5
LOLA significantly decreased blood ammonia and improved hepatic function (p<0.05 or 0.01). LOLA is effective for hepatic encephalopathy and has not obvious side effect.
Therapeutic effects of LOLA in HE patients
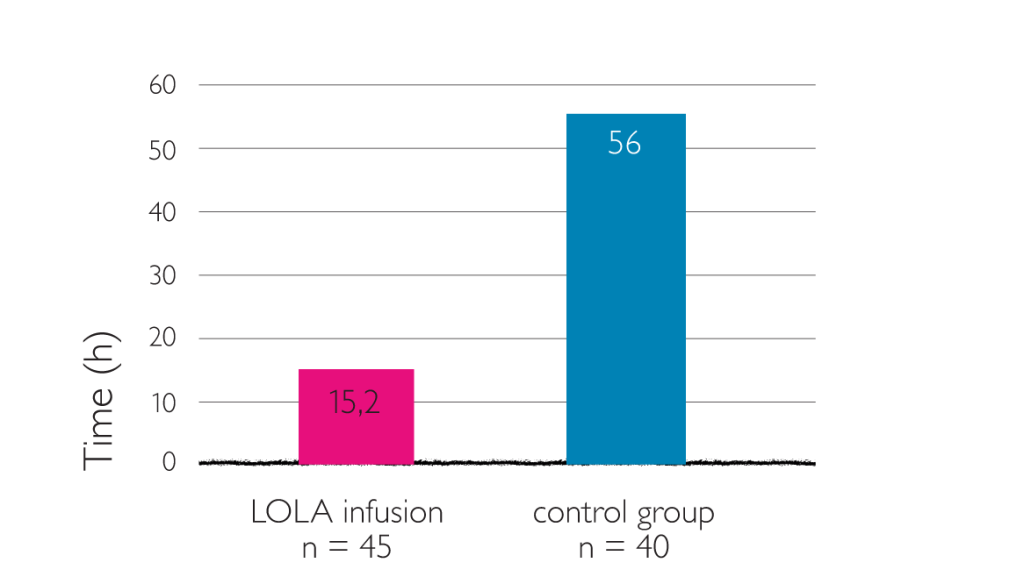
Average time for complete recovery of consciousness (LOLA once daily, 40 ml in 250 ml 10% glucose and saline)5
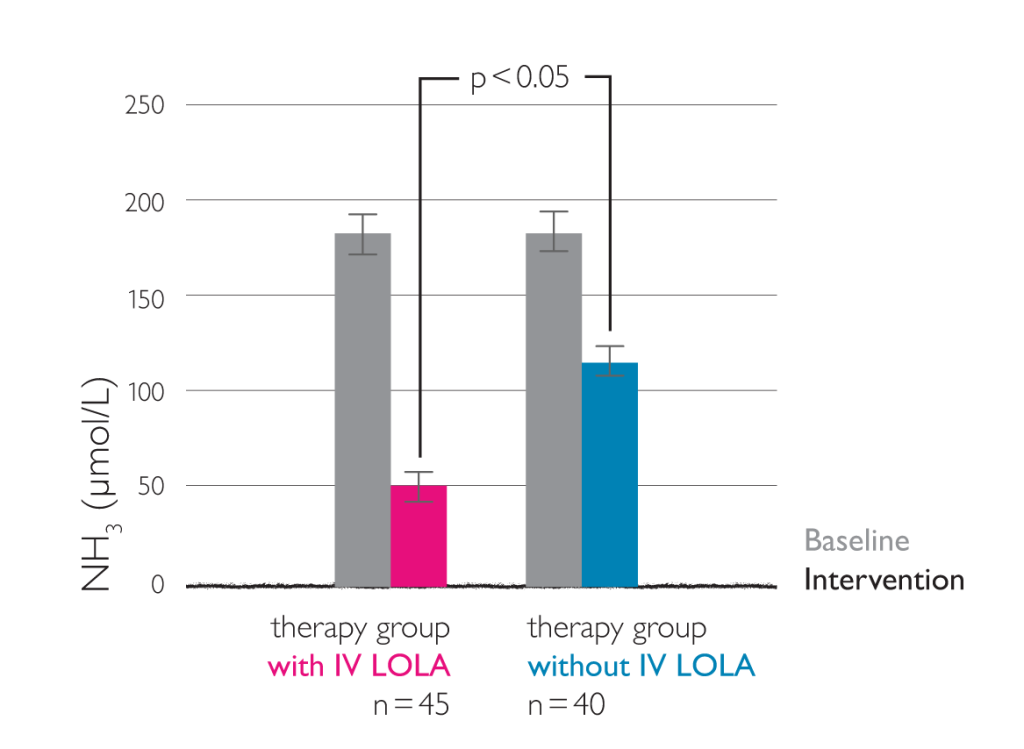
Changes in blood ammonia (LOLA once daily, 40 ml in 250 ml 10% glucose and saline)5
Efficacy of oral Hepa-Merz®
The oral form of LOLA is important for long-term treatment and maybe used for treatment of associated conditions and sequelae of diseases with reduced detoxification functions of the liver.
Oral L-ornithine-L-aspartate therapy of chronic hepatic encephalopathy: results of a placebo-controlled double-blind study. Stauch S et al. 1998.6
Placebo-controlled double-blind trial on 66 patients with cirrhosis of the liver and HE grade 0 to II. All patients had to have demonstrable hyperammonemia (fasting venous ammonia concentration >50 µmol/l) and a performance time >30 s in Number Connection Test (NTC-A).
Treatment with LOLA was associated with significant improvements:6
- reduction of postprandial venous ammonia concentrations (p<0.01)
- decrease of ammonia concentrations under fasting conditions
- reduction in performance time for the NTC-A (p<0.05)
- improvement of HE grade after 14 days of LOLA treatment (p<0.05)
Evidence for the efficacy of LOLA in treating HE patients
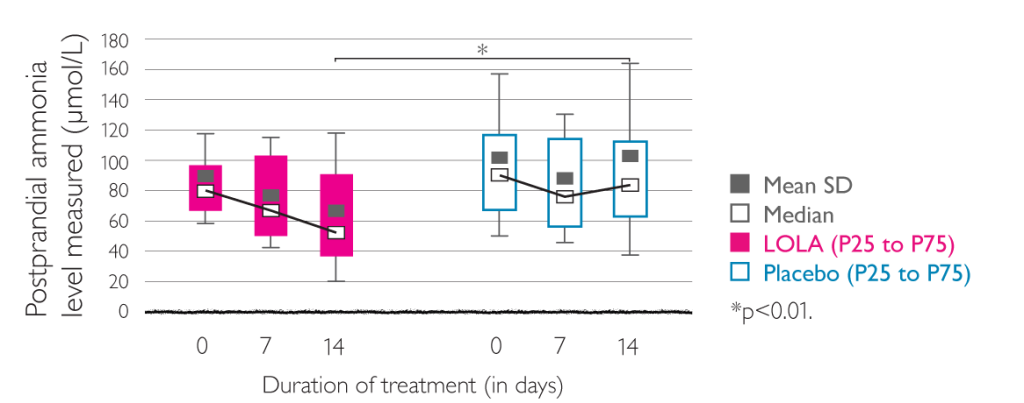
Effects of LOLA (left) and placebo (right) on postprandial venous ammonia concentration. Figures given are the mean (black square) and standard deviation, median (white square), and 25th and 75th percentiles.6
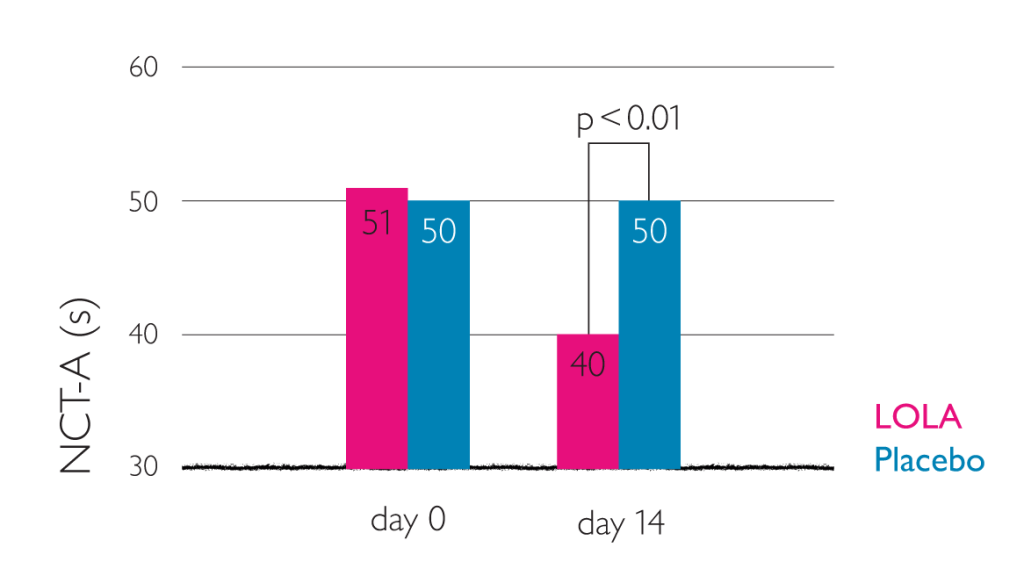
Reduction in NCT-A performance time (administration of 3 x 6 g oral LOLA)6
These placebo-controlled clinical trials showed that the oral form of LOLA is comparable to intravenous LOLA with respect to efficacy and tolerability.6
Post marketing surveillance study on effectiveness and tolerability of oral LOLA. Grüngreiff K et al. 2001.7
Multicentre (n=250) post-marketing surveillance involving 1167 patients with fatty liver, chronic hepatitis, cirrhosis, increased transaminases, and clinical symptoms. Patients were treated with oral LOLA at freely selected dosages and for freely selected durations of treatment. Measures included clinical symptoms (tiredness, HE, chemical laboratory parameters (γ-GT, ASAT, ALAT, bilirubin, Quick). Three dosage groups receiving equivalent to < 9 g LOLA / day, = 9 g LOLA / day or > 9 g LOLA / day.7
Oral LOLA in chronic liver disease:7
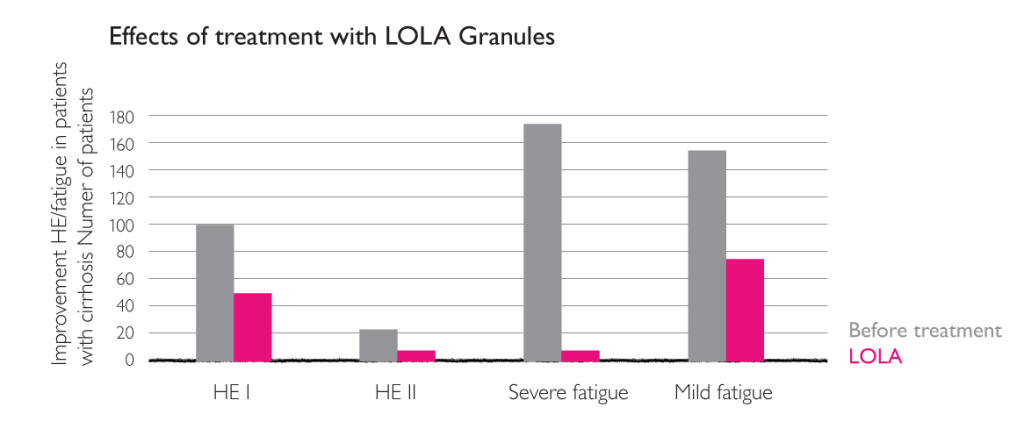
Effects of treatment with LOLA granules on the clinical parameter “fatigue”.7
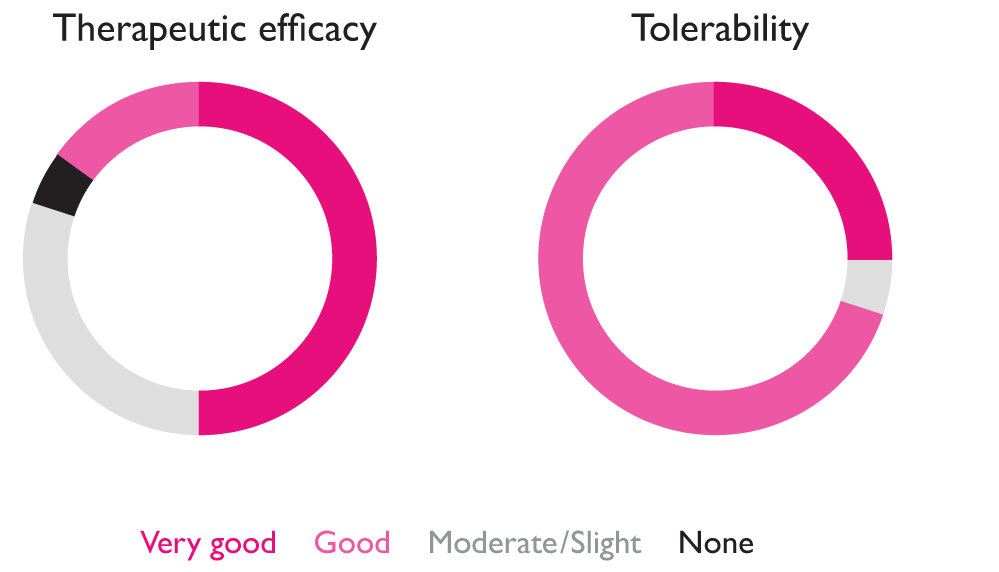
Overall assessment of the efficacy (left) and tolerability (right) of treatment with LOLA granules at the end of treatment, shown as percentages.7
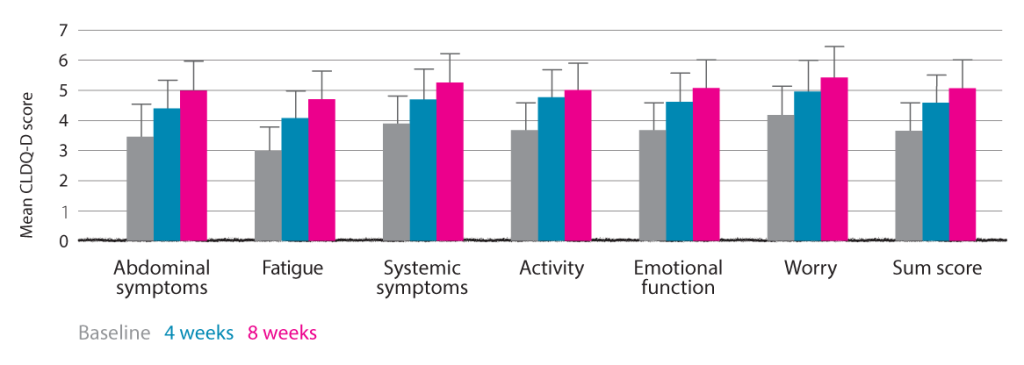
Assessment of HRQOL measures during eight-week treatment with LOLA. Self-assessment by patient using German-validated version of Chronic Liver Disease Questionnaire (CLDQ-D).7
The results of experimental clinical studies are in agreement with findings in experimental animal studies. The clinical efficacy and tolerability of LOLA infusions, with oral administration, or a combination of the two, have been investigated in patients with mild to severe impairment of hepatic function, including cirrhosis, where duration of treatment ranged from a few days to several years.
With respect to its efficacy in cirrhosis and HE ranging from MHE to grade II, treatment with LOLA in comparison with placebo shows that:
- the mental state (HE grade) significantly improves
- the PSE index improves significantly
- performance time in NCT-A is significantly reduced
- venous ammonia concentrations in the fasting and postprandial states are significantly lowered
Placebo-controlled clinical trials show that, with respect to efficacy and tolerability, the oral form of LOLA is comparable to LOLA infusion concentrate for the treatment of HE.
Meta-analysis of LOLA efficacy
Three randomised placebo-controlled trials with 277 patients:
- LOLA treatment for at least 7 days
- Patients with cirrhosis and HE
Meta-analysis of these three trials showed a significant improvement of patient’s mental state after treatment with LOLA. The improvement in mental state is already significant and important. In HE the clinical outcome of the patient is more helpful in determining whether the patient’s status is improving or not.8
